This article is the third in a series of posts on clean hydrogen’s role in building a net-zero future. Other articles in the series can be found below:
It’s not just production and usage that we need to consider—how we store and transport hydrogen impacts both emissions and costs.
Deep underground in Delta, Utah, two giant empty salt caverns are getting a makeover. Large enough to store 4.5 million barrels of oil, these vast empty spaces are being converted into the nation’s largest clean hydrogen storage facility. Called the Advanced Clean Energy Storage Hub, it’s poised to demonstrate the scale and promise of geologic (underground) hydrogen storage.
We’ve already talked about the public and private sector investment pouring into hydrogen and various ways to produce and use hydrogen. In this post, I’ll focus on storage and transport, two equally important—but often overlooked—steps in the hydrogen system. Because it’s not just production and usage that we need to consider—how we store and transport hydrogen impacts both emissions and costs.
I like to think of clean hydrogen as a choose-your-own adventure.
It is hydrogen’s flexibility as an energy carrier that makes it such a powerful tool in our decarbonization toolkit. It’s especially valuable where no other affordable alternatives exist, such as industrial processes that you can’t easily electrify or where you need it as a feedstock. Solving for the lowest emission, lowest cost way to get hydrogen to those uses means you have to consider how you use it, how far away you’re using it from where it’s produced, and how long in the future you’ll need to use it. Those factors will dictate how it’s transported and stored, and by extension, what form it takes on that journey from production to use.
There are multiple options for building the middle section of this journey, or “value chain”, illustrated in simplified form below (to see a more thorough schematic, check out Figure 4 here).
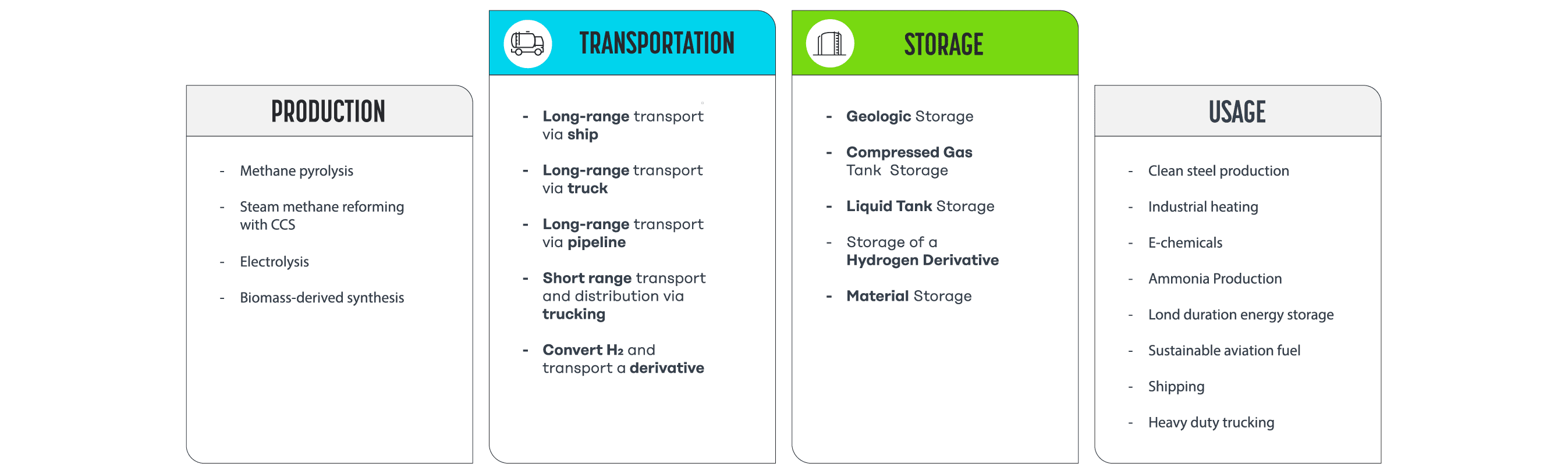
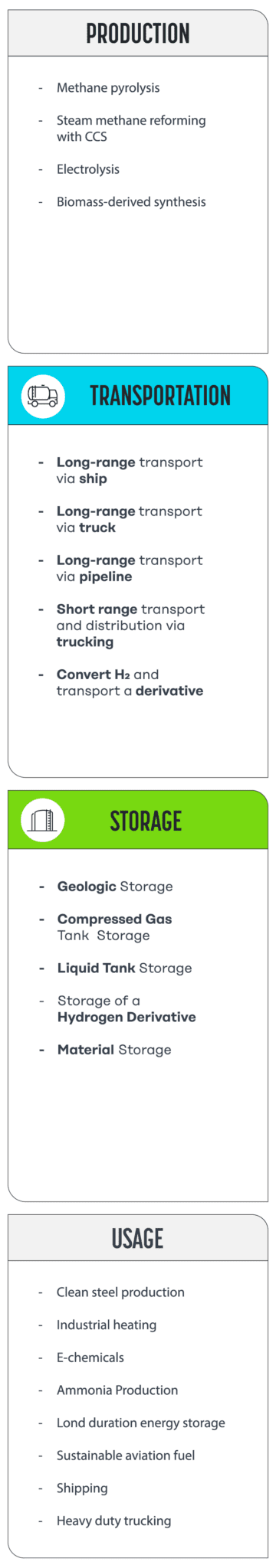
As you can see, options for transport and storage can require changing the physical state of the hydrogen from a gas to a liquid or solid, compressing it, or chemically converting it to another carrier. These transformations can make it easier to contain and move hydrogen because generally speaking, as a gas, hydrogen is lighter than air and not very energy dense by volume. Yet, transformations, transport, and storage aren’t without tradeoffs, as every new step or form of complexity in the process adds cost. Let’s dive into the options.
There are a number of ways to transport hydrogen.
Trucks
Hydrogen can be transported by truck one of two ways: via a liquid tanker or by a “tube trailer” with compressed gas cylinders. Trucking is a flexible option for supplying hydrogen to regions where demand is still developing. It’s also how laboratories and other facilities that require smaller volumes receive hydrogen today. However, only a limited volume can fit on a single truck and the trucks of course require fuel of their own to move the hydrogen itself.
Pipelines
Pipelines are best suited for delivering clean hydrogen to end users with large and steady demand, like a steel plant, over long distances. While we don’t know yet how much pipeline capacity we’ll need to support hydrogen’s use as a decarbonization tool in the U.S., we do know that repurposing existing natural gas infrastructure for hydrogen is nontrivial because hydrogen tends to degrade pipes not specifically designed for it. However, either way, the IEA suggests that once operational, pipelines are a cost competitive way to transport hydrogen at long distances up to 5000 km.
Ships
Over even longer distances, moving hydrogen in some liquid form (more on that in a bit) by ship is likely economical. The world’s first long distance shipment of liquid hydrogen was completed last year, potentially the first step in building a global clean hydrogen supply chain. Costs and energy use remain high, and it’s far from certain we’ll be shipping hydrogen in pure form; the International Renewable Energy Agency estimated that nearly half of hydrogen traded between countries in 2050 would instead be in the form of ammonia.
There are still lots of questions about how far we’ll need to transport hydrogen, and in what form. But in the short term, as we learn from the first efforts to use hydrogen in new ways, keeping hydrogen production and use close is better at this early stage because it uses less energy and costs less money. That’s why the Energy Department’s strategy for scaling hydrogen focuses on regional hubs. At the end of the day, we’re likely going to see a combination of all of the above.
There are four main ways to store hydrogen.
Geologic Storage
Hydrogen can be stored as a gas underground in empty salt caverns, depleted aquifers, or retired oil and gas fields. In fact, there’s a long precedent of storing gasses underground like this. Doing so is called ”geologic” storage, and it’s an ideal option for storing hydrogen for long periods of time, as is needed for seasonal energy storage. It’s one of the cheapest and largest scale options today, but it’s not available everywhere.
Compressed Gas
Like any gas, hydrogen can be compressed and stored in tanks. But hydrogen requires very high pressure tanks that hold a limited quantity of energy. Whether we’re talking about above ground tanks or tube trucks, compressed gas is one of the most expensive and least energy dense options we have today, but it’s also one of the simplest.
Liquid and Cryogenic Storage (Or storage in another phase)
Hydrogen is much more energy dense as a liquid. The downside is that getting there means cooling it to near absolute zero, requiring significant energy and sophisticated insulated tanks. Historically, this has only made sense for space travel as rocket fuel, but could be used for long distance shipping.
Breakthrough Energy Fellows team, Verne, is developing a method to store hydrogen as a cold, compressed gas (“cryo-compressed”) making high-density hydrogen more economical for heavy-duty transportation.
As mentioned above, hydrogen can also be converted into liquid carriers like ammonia, which has a higher energy density and a developed global infrastructure because it’s already used for fertilizer and chemical production.
Material Storage
Hydrogen can also be chemically bonded to materials that pack hydrogen gas more densely through a process called adsorption, though we need more R&D to understand cost and scalability.
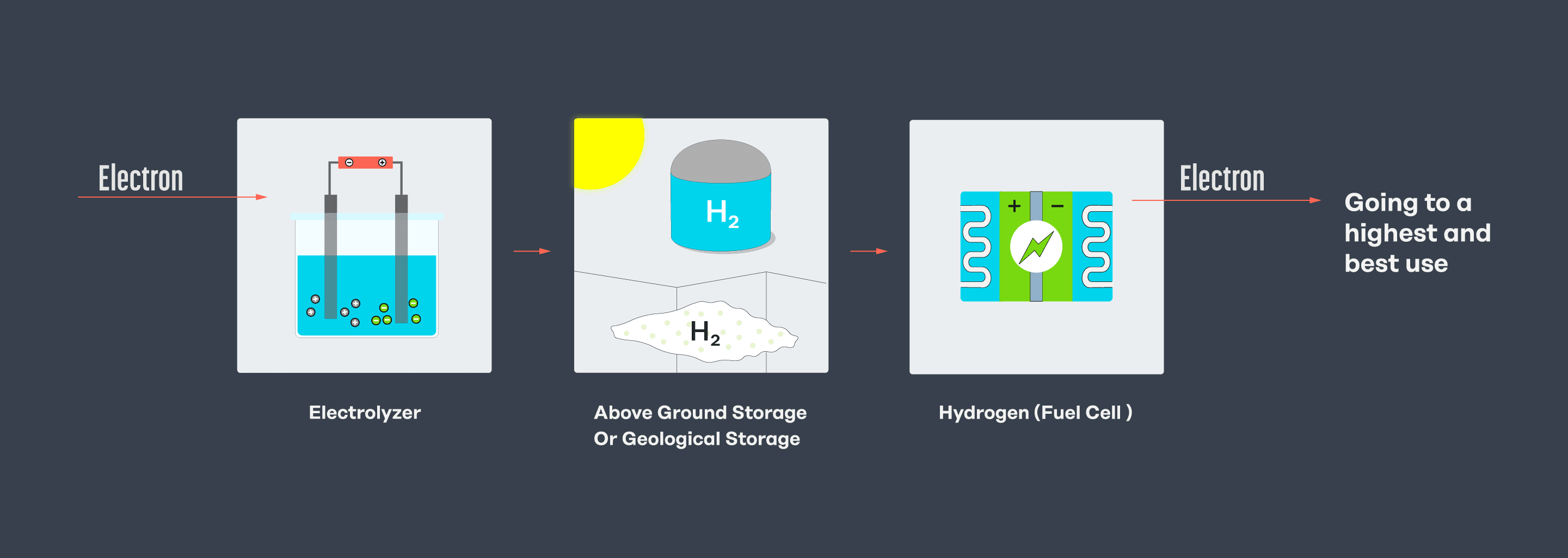
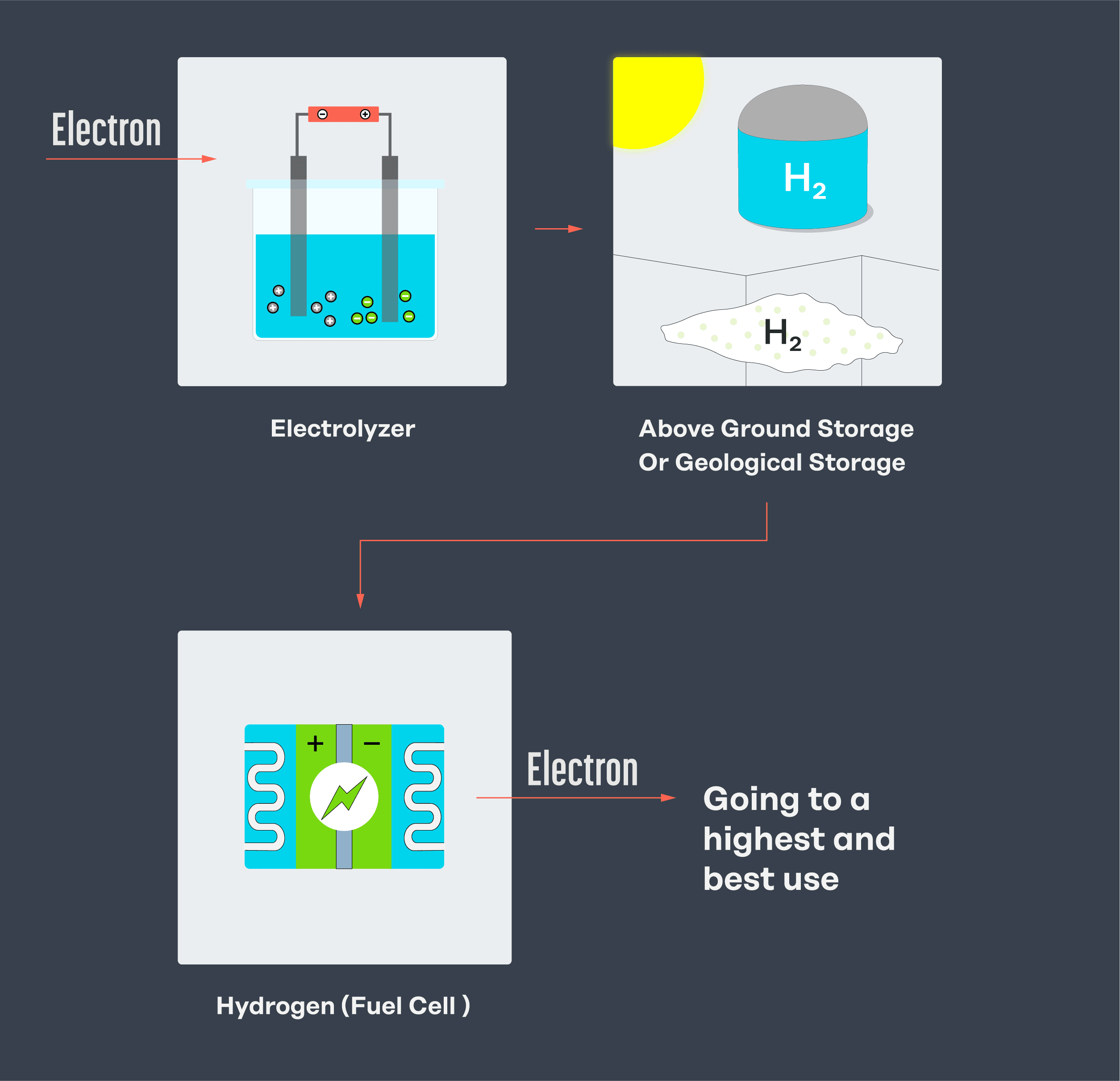
Below is a simplified sketch of what it would look like to turn electricity into hydrogen, store it, and then reconvert it to electricity. Turning electricity into hydrogen only to make electricity again is inefficient, so doing so only makes sense in a few applications – like seasonal energy storage or fueling a fuel cell, such as for heavy transport and aviation.
Hydrogen poses a few big challenges.
The transport and storage options above will keep getting cheaper and more efficient as we do them more. But this marvelous molecule and the complex systems it requires both pose unique challenges.
For one, hydrogen behaves differently than other fuels. It’s more flammable than natural gas—and the flame is nearly invisible, requiring special sensors.
And because hydrogen atoms are so small, they can permeate certain materials, making them brittle and causing cracks and leaks. That makes it vitally important to select the right materials for tanks and pipelines. However, there are best practices that enable the current industry to use hydrogen safely. We just need to ensure that new protocols are developed to cover new use cases.
In addition to maximizing safety and minimizing risks, we need to study more about hydrogen leakage and its climate impacts. Hydrogen can indirectly act as a greenhouse gas if released into the atmosphere by extending the lifetime of methane emissions, and right now we don’t have good data on how much hydrogen leaks, and from where. Despite this, analysis shows that using clean hydrogen instead of fossil fuels results in much lower overall emissions in the short term, even if hydrogen leakage occurs, making it the better choice (for the nerds who want to see data, this figure shows how emissions associated with hydrogen are much lower than fossil fuels, and how they quickly drop to zero – the emissions associated with “blue” hydrogen here include emissions from leaks of methane used to produce the hydrogen).
The bottom line:
Policymakers, project developers, and advocates should consider every stage of the hydrogen process and strike a balance between what’s cheaper and what has the highest decarbonization potential. The optimal solution may vary by project and region. R&D is progressing at a breakneck pace and our knowledge is improving rapidly, so the decisions we make today might be different than a decade from now.
In my next post, we’ll explore how the entire hydrogen lifecycle—how we produce, move, transport, and use hydrogen—impacts its potential as a decarbonization tool.

